It happened.
Exactly as predicted.
Unanimous vote “for all vaccines administered currently [to] be a bivalent [booster] vaccine (Original Plus Omicron BA.4/BA.5).”
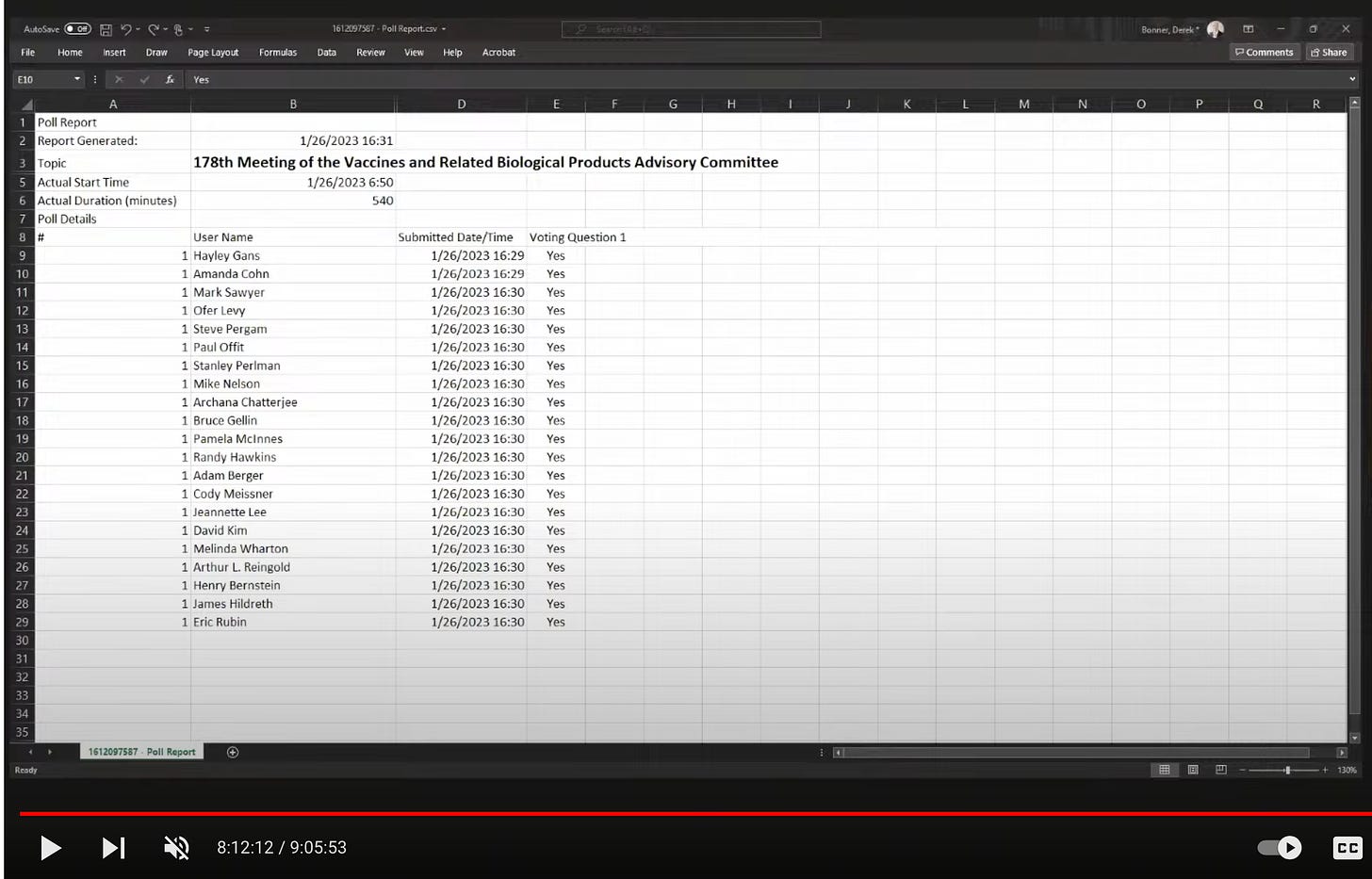
Here’s a screenshot of the voting question:
And the votes:
You can watch the whole nine-hour meeting here. The voting process starts around the 7:42:00 mark. Let’s dig into it.
Timeline of Events Leading Up to the Vote:
Jan/Feb 2022 - Pfizer and Moderna produced their versions of bivalent booster vaccine.
28 June 2022 - “Researchers from Pfizer–BioNTech and Moderna presented data on their [EUA] bivalent [booster] vaccines to the FDA’s Vaccines and Related Biological Products Advisory Committee… the results were underwhelming” - Dr. Paul Offit.
29 June 2022 - “A series of rapid-fire policy decisions followed. On June 29, 2022, the day after the advisory committee meeting, the Biden administration agreed to purchase 105 million doses of Pfizer–BioNTech’s [EUA] bivalent [booster] vaccine containing BA.4 and BA.5 mRNA.” - Dr. Paul Offit. HHS Press Release
29 July 2022 - “One month later, on July 29, 2022, the administration agreed to purchase 66 million doses of Moderna’s [EUA] bivalent [booster] vaccine, intending to offer both vaccines in the fall and winter.” -Dr Paul Offit. DoD Press Release
*HHS & DoD spent a combined $4.94 billion on 170 million Emergency Use Authorized (EUA) bivalent boosters (with option to expand up to 600 million doses).
1 September 2022 - The FDA withdrew its emergency use authorization for monovalent vaccine boosters.
2 September 2022 - The CDC began recommending EUA bivalent boosters.
18 September 2022 - President Biden declares the Pandemic is over on 60 Minutes.
21 September 2022 - Press Secretary Karine Jean-Pierre walks back President’s “Pandemic is Over” declaration.
13 October 2022 - HHS Secretary Becerra Renews the Covid-19 Emergency Declaration for the 11th time.
10 January 2023 - SECDEF Austin rescinds DoD Mandate.
11 January 2023 - HHS Secretary Becerra Renews the Covid-19 Emergency Declaration for the 12th time.
11 January 2023 - Dr. Paul Offit Published “Bivalent Covid-19 Vaccines — A Cautionary Tale” in the New England Journal of Medicine (DOI: 10.1056/NEJMp2215780).
26 January 2023 - FDA’s VRBPA Committee unanimously voted (including Dr. Offit) that all covid vaccines would be the EUA bivalents— as primary and boosters.
What Do We Know?
We know that the bivalents were produced early 2022. We know that U.S. Government spent nearly $5 billion on bivalents in the summer of 2022. The first purchase being made the day after FDA voted to approve the bivalents as boosters. We know the bivalents are Emergency Use Authorized. Therefore, legally speaking, there needs to be an Emergency to justify using EUAs. We know, that the emergency was renewed twice after the $5 billion purchase. What else do we know?
Bivalent Acceptance Rates
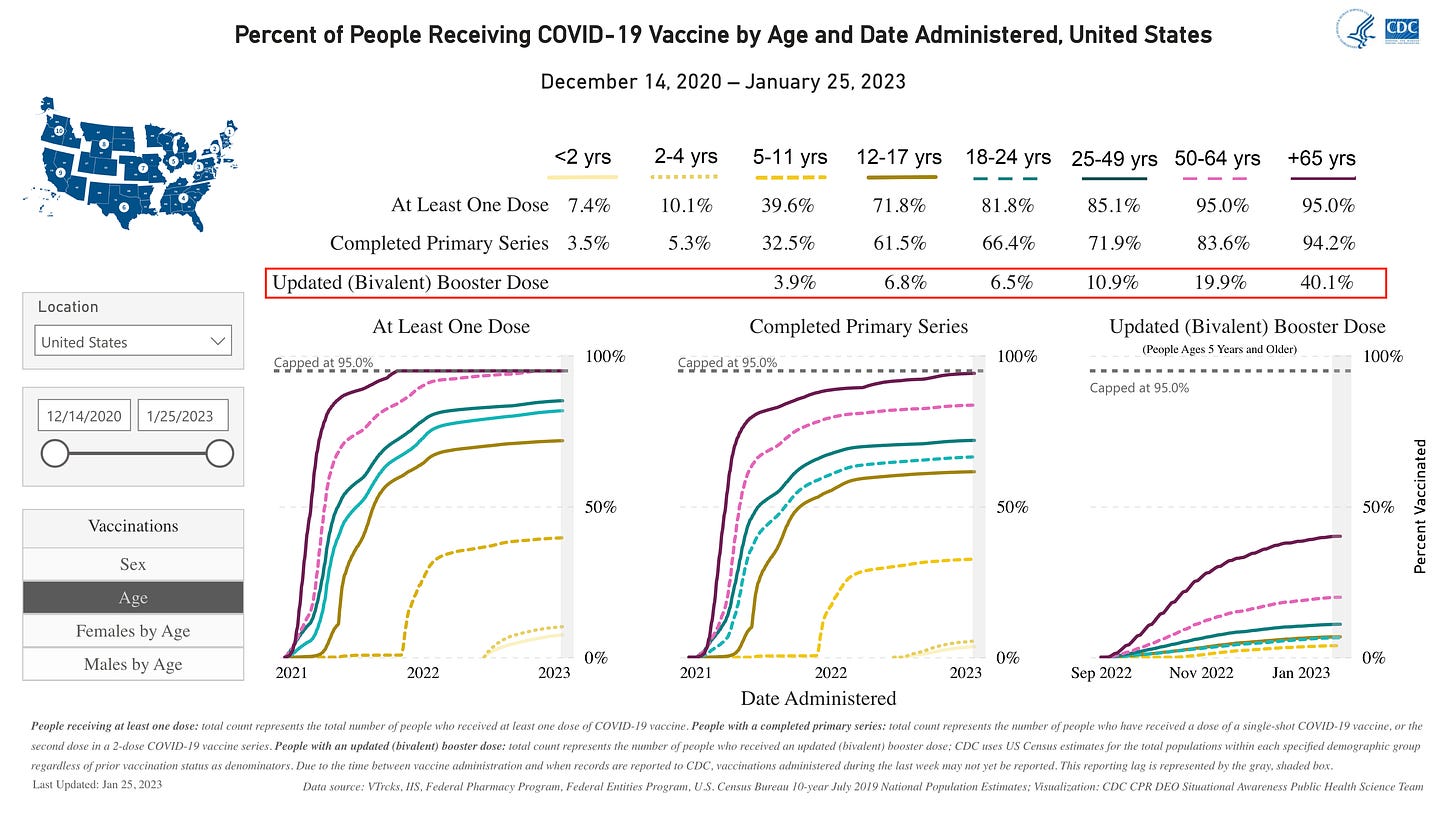
According to the CDC, bivalent boosters are essentially sitting on the shelf waiting to expire.
In nearly all age groups the acceptance rate is 11% or lower. The first notable uptake is 50-64 age group at 20% acceptance. Followed by 65+ with 40% acceptance.
Dr. Offit is aware of this dilemma. He cites the statistics from November: “As of November 15, 2022, only about 10% of the population for whom the bivalent vaccine had been recommended had received it.”
Keep in the mind that the bivalents became available in September of 2022, nearly 5 months ago. The general public is simply not interested in getting boosters.
Are Bivalents effective?
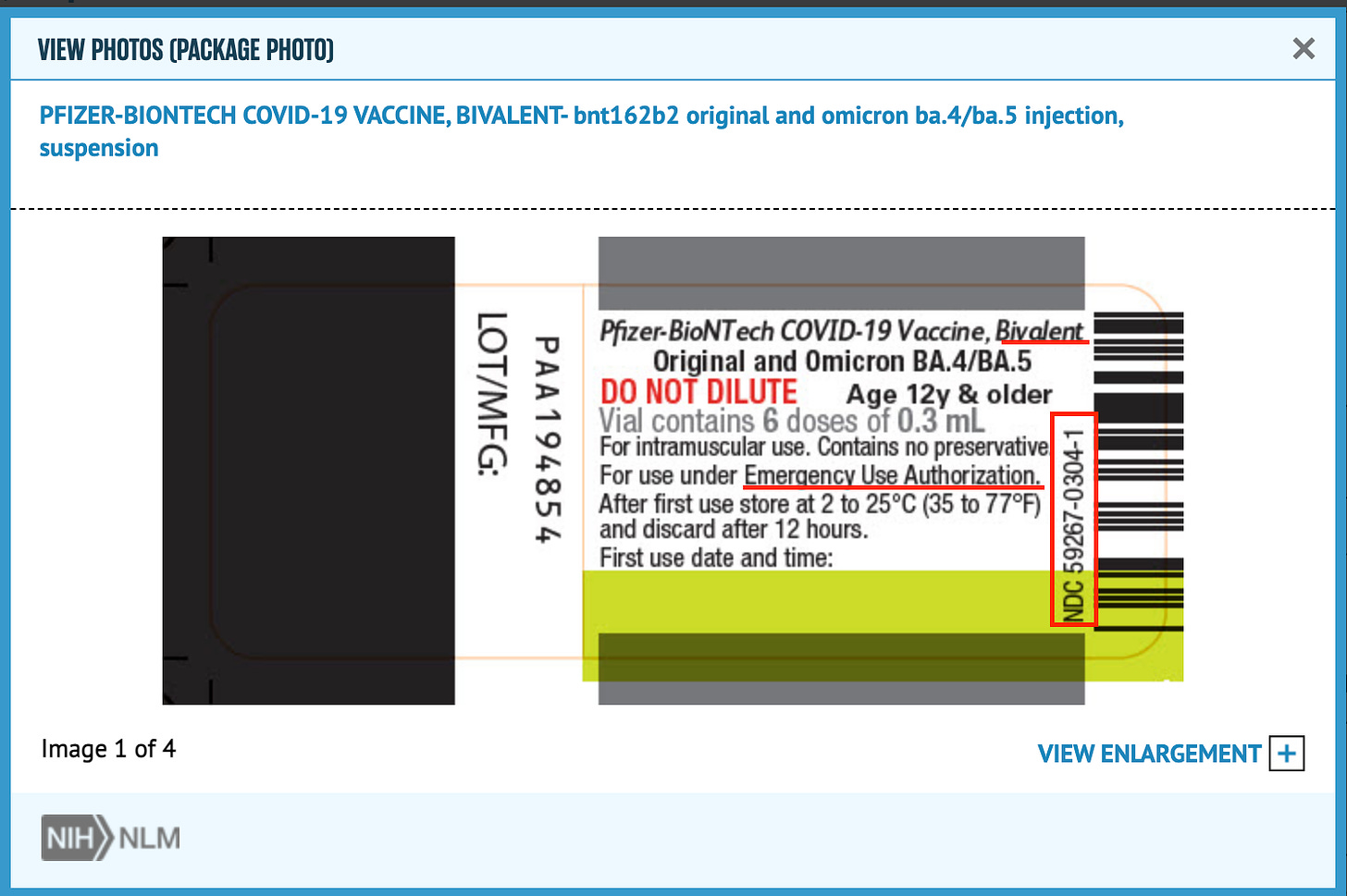
We know the bivalent boosters were developed to protect against the original strain, BA.4, and BA.5. It’s on the label:
Dr. Offit explains: “The [June 28, 20222] FDA advisory committee, sensing the urgency of responding to these immune-evasive strains, voted to authorize bivalent vaccines with an understanding that they would target omicron subvariants BA.4 and BA.5, which at the time had accounted for more than 95% of circulating strains.”
Today it’s a much different story. The strains that the bivalents were designed to protect against are virtually non-existent.
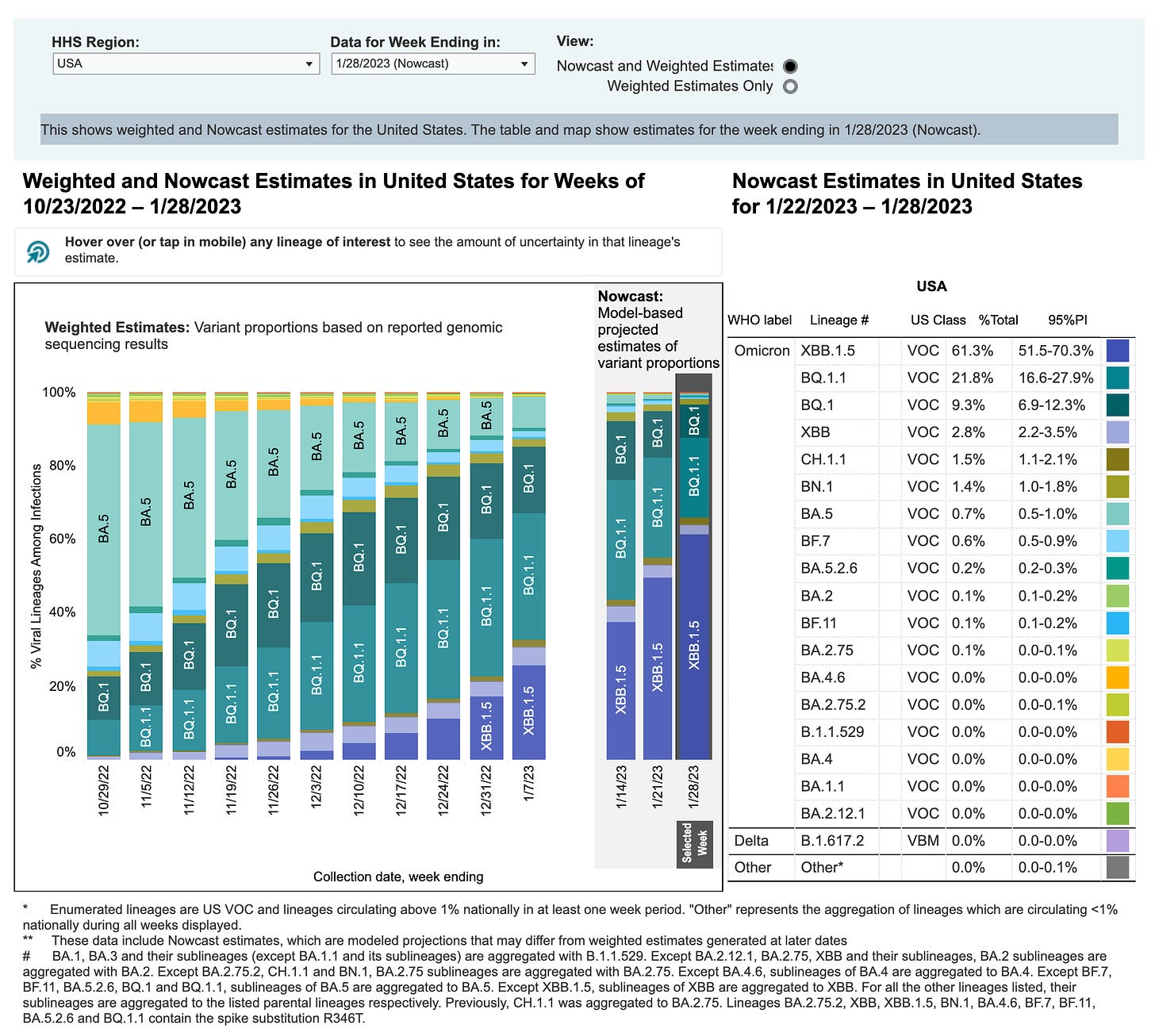
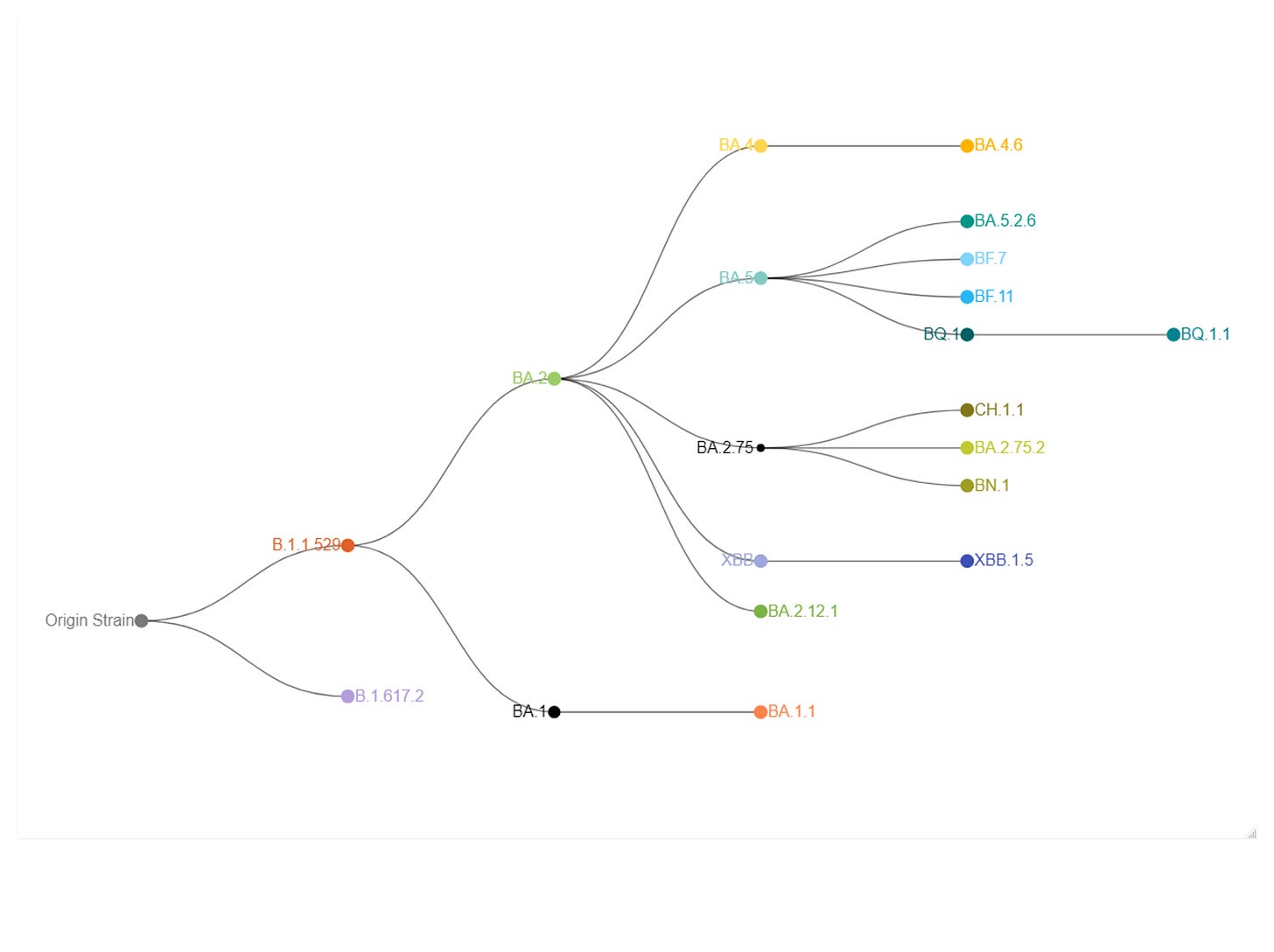
Here’s the CDC data for week ending in Jan 28th, 2022:
The Original, BA.4, and BA.5 strains make up only 0.7% of the current circulating strains.
Let’s give the FDA the benefit of the doubt and include all the sub-variants of BA.4 and BA.5 for good measure. (*Note; This is just a thought experiment, it doesn’t work like this but let’s just say it does for argument’s sake).
To include the sub-variants means we would calculate: BA.4 (0.0%); BA.4.6 (0.0%); BA.5 (0.7%); BA.5.2.6 (0.2%); BF.7 (0.6%); BF.11 (0.1%); BQ.1 (9.3%); BQ.1.1 (21.8%).
Tallying those variants gives us a total of: 32.7% with the BQ.1 and BQ1.1 mutations making up the majority at 31.1%. Assuming the bivalents would offer protection against these strains, the recipient is still largely exposed to the constantly mutating variants.
As a non-virologist, I have to ask another set of questions:
When were the Bivalents developed?
When did BQ.1 and BQ.1.1 mutations occur?
What data is available to ensure the bivalents are effective against B.Q.1 and BQ.1.1?
Answers:
We know the bivalents were produced in Jan/Feb of 2022. The data was then presented on June 28th 2022. Biden Administration began purchasing bivalents the next day. Within a few months, the CDC started recommending bivalents the day after FDA withdrew the EUA for monovalent boosters. That was evident per the timeline. In addition here’s when the CDC, had the updated (bivalent) boosters available:
September 2, 2022, for people aged 12 years and older
October 12, 2022, for people aged 5–11 years
December 9, 2022, for children aged 6 months–4 years who completed the Moderna COVID-19 vaccine primary series
BQ.1 and BQ.1.1 started appearing November of 2022. WebMD dates them around Nov 13, 2022. Nearly 10 months after the bivalents were produced.
Not much. However, Dr. Offit’s article published on 11 Jan 2023 gives us some insight on how effective the bivalents were against BA.4 and BA.5.
According to Dr. Offit, two separate research groups determined that the bivalent boosters do not elicit superior immune responses.
Qian Wang, et al. found “Boosting with new bivalent mRNA vaccines targeting both the BA.4–BA.5 variant and the D614G strain did not elicit a discernibly superior virus-neutralizing peak antibody response as compared with boosting with the original monovalent vaccines.”
A.Y. Collier et al. found that “BA.5 neutralizing antibody titer was similar after monovalent and bivalent mRNA boosting.”
In other words the bivalents made no difference.
Summary
The Bivalents were unanimously voted to replace all vaccines currently administered in the United States on Jan 26th, 2023. The CDC has not changed their guidelines.
Dr. Offit, two weeks prior to the VRBPAC, published an article criticizing the bivalents. Yet on voting day, he voted in unison with the rest of the members on the Committee.
In Dr. Offit’s paper he claims that the Bivalents did not show significant increase in protections against BA.4 and BA.5. There is limited data on booster effectiveness against the new dominate variants.
Today, the strains the bivalent booster was developed to counter are virtually non-existent—making up 0.7% of the current circulating strains.
The bivalent boosters are unlicensed emergency use authorized.
Per Title 21 U.S. Code § 360bbb–3; there must be a declared emergency before HHS can grant an emergency use authorization.
HHS renewed that emergency declaration on January 11th, 2023 for the 12th time, the same day Dr. Offit’s paper is published critiquing the EUA bivalents. Coincidentally, this was the same day that the entire U.S. Federal Aviation industry was grounded due to the NOTAM system failure.
Conclusion
The FDA decision was not a science based decision. It was an administrative decision with the groundwork being laid out by multiple actors over the course of a year. Pfizer created them. FDA approved them as boosters. HHS and DoD purchased them. HHS continued to renew the emergency. FDA voted to replace all vaccines with the bivalents—while one of their own criticized the effectiveness.
Meanwhile, the variants have mutated beyond the bivalent boosters capability and the public is not interested in receiving them.
Do not misunderstand me. I am not advocating for better more up-to-date vaccines. Quite the opposite. My point is that the entire pandemic response has been about Executive Overreach rather than the safety of the public. The FDA vote to have the outdated emergency use bivalents replace all the covid vaccines is a prime example.
I would not be surprised if the bivalents miraculously receive an approval in the near future.



the entire alphabet agencies and nearly all the government are bought and paid for.
Thank you for the thorough article.